Enzyme Lab
Background:
Enzymes mostly come from proteins and are used as catalysts to speed up chemical reactions. These catalysts lower the activation energy needed and decrease the time it takes for the reaction to occur. The shape of the activation site located on the enzyme fits with a specific substrate or reactant similar to a lock and key fit. The allosteric site of the enzyme is responsible for keeping the shape through cofactors. If the pH level or temperature is changed to an extreme, the enzyme could experience denaturation which prevents any continuing repetition of the enzyme's job. In this lab, enzymes from plants will be used to speed up the separation of oxygen gas and water from a compound solution of hydrogen peroxide.
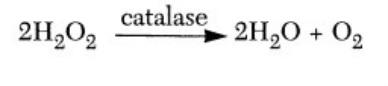
This type of enzyme used to decompose oxygen and water is most commonly known as catalase. Catalase is an enzyme exposed to oxygen found in most life. If hydrogen peroxide is added to a tissue or substance containing catalase, the combination will result in bubbling. When an organism is undergoing metabolism hydrogen peroxide often becomes a product which is known to damage cells. Catalase enzymes work in order to decompose the hydrogen peroxide into safer substances such as water and oxygen.
Enzyme-Substrate Equation:
E + S ⇌ ES → E + P
Interaction between hydrogen peroxide and catalase:
This type of enzyme used to decompose oxygen and water is most commonly known as catalase. Catalase is an enzyme exposed to oxygen found in most life. If hydrogen peroxide is added to a tissue or substance containing catalase, the combination will result in bubbling. When an organism is undergoing metabolism hydrogen peroxide often becomes a product which is known to damage cells. Catalase enzymes work in order to decompose the hydrogen peroxide into safer substances such as water and oxygen.
Enzyme-Substrate Equation:
E + S ⇌ ES → E + P
Interaction between hydrogen peroxide and catalase:
Pre-Lab:
For the Pre-Lab, two carrot pieces and two green bean pieces were immersed in hydrogen peroxide to observe the enzyme catalyst reaction changing the compound to water and oxygen gas.
Hypothesis: When carrot tissue is added to hydrogen peroxide, the compound will turn into oxygen and water faster than the green bean tissue.
Independent Variable: The amount of the enzyme catalase in the vegetable.
Dependent Variable: The rate of the catalase reaction that decomposes the hydrogen peroxide.
Dependent Variable: The rate of the catalase reaction that decomposes the hydrogen peroxide.
Enzyme Lab Set Up:
For the Enzyme Lab, two 1 gram pieces of a turnip were added to a reaction chamber filled with 10 mL of hydrogen peroxide. The chamber shown below is attached to a lid with a dropper that has an opening which allows the oxygen gas created by the enzyme reaction between the turnip pieces and hydrogen peroxide to escape into the graduated cylinder. When the the oxygen gas enters the cylinder water is pushed out of the cylinder and into the bath with the oxygen remaining at the top of the cylinder. As the Oxygen flows through the graduated cylinder, the water inside decreases at a constant rate.
Data Table:
Initial Rate of an Enzyme:
The initial rate of an enzyme defines the speed of the enzymes reactions with chemicals. The first interval of time is usually used to calculate this. In this experiment, the initial rate was taken from the 30 and 240 second intervals.
(m2-m1)/(t2-t1) = (1 mL - 0)/(30 secs - 0) = 0.033
(10 mL - 0)/(240 secs - 0) = 0.0417
(m2-m1)/(t2-t1) = (1 mL - 0)/(30 secs - 0) = 0.033
(10 mL - 0)/(240 secs - 0) = 0.0417
Summary:
The initial rate of the turnip's enzyme indicates that the time for decomposition of hydrogen peroxide is less than if the enzyme was not added. This proves that catalase is a significant factor in the breakdown of hydrogen peroxide. When the initial rate of 1 mL of oxygen gas at the 30 second interval is compared to the rate of 10 mL after 240 seconds (4 minutes), the trend of the decomposition seems to have increased in rate. As seen in the graph of data above, the overall trend of oxygen in the cylinder increases at a linear rate. This rate will probably continue until the whole cylinder is filled with oxygen gas and the water has escaped into the bath. If a variable is added to the experiment such as heat or an acid, the initial rate of oxygen collected in the graduated cylinder will be significantly less than the initial rate of the original experiment with the catalase. In the experiment, another way to determine the enzyme activity would be to look at the displacement of water in the test tube. In reality, determining enzyme activity within the body include pouring hydrogen peroxide on tissue containing catalase. This will result in bubbling of the tissue.
Sources of Error:
Possible sources of error include recording oxygen gas accumulation in nonperfect intervals of 30 seconds, the cap of the reaction chamber was not placed quickly enough, and the reaction chamber was not submerged in the water in a timely manner. If the experiment was to be repeated, these sources of error could be prevented by actively paying attention to time and accuracy in placing the components of the experiment set up.